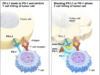
A group of researchers from the Johns Hopkins Kimmel Cancer Center reported a successful clinical trial of using the immunotherapy drug nivolumab (anti-PD-1) on patients who did not respond to traditional treatments initially. The trial gave these patients, who were being treated for melanoma, renal cell carcinoma (RCC), or non-small cell lung cancer (NSCLC), multiple doses of anti-PD-1.
The Study Participants and Findings
The study was conducted on just under 300 adults of both sexes. The most favorable part of the study was the increased five-year survival rate of patients who received immunotherapy, which was more effective than other treatments like chemotherapy, biological therapy, or antiangiogenic therapy. Before the trial, the three-year survival rates for melanoma patients was 5% and the five year survival rate for NSCLC was 6%. But when patients were given anti-PD-1 immunotherapy, the survival rates increased to 34.2% and 15.6% respectively.
Most scientists think this study is long overdue. Anti-PD-1 has been known to enhance the cancer killing ability of T-cells for a long time, but there has never been a long term clinical outcome study.
How Does Anti-PD-1 Work?
Immune cells have a molecule called PD-1 that help it to fight disease. However, cancer cells can hide from this molecule so the body does not fight them. Anti-PD-1 makes it so the PD-1 molecule can see the cancer again and begin to attack. Much of the research and development behind these drugs has been accomplished by the Bloomberg Kimmel Institute at Johns Hopkins. The FDA has approved anti-PD-1 to fight against multiple types of cancer.
Why These Patients Were Chosen
The study began in 2008 and encompassed patients with three different types of cancer from more than a dozen U.S. medical centers. Patients received different doses of the drug every two weeks for two years. All the patients had advanced cancer, meaning cancer that had metastasized and spread to distant parts of the body, and all had previously been treated by multiple traditional therapies. Almost half of the patients had undergone more than three other therapies.
The researchers, however, said the number of prior treatments did not affect the five-year survival rate increase in any significant way. The study also reported lower survival rates for patients whose cancer had spread to the bones or liver and patients with low immune cell counts. The patients who had cancer but did not exhibit cancer symptoms had a much higher survival rate.
The Future of Anti-PD-1
The research team is hopeful their study can guide doctors and patients on how to use anti-PD-1 effectively. This trial establishes that the drug is safe for use, and can now be applied to other cancers in more situations. Some patients may even receive anti-PD-1 as their first form of treatment.
This team of researchers also previously reported that there were more favorable responses to surgery for NSCLC when anti-PD-1 was given to the patient before the surgery. This could mean that cancer drugs will be used earlier in the treatment process than they were before.
One of the most surprising things about the study was that patients who had side effects caused by the anti-PD-1 drug survived almost a month more on average than patients with no side effects. Roughly 15% experienced adverse side effects like fatigue and rash, but these effects can be managed so they are not very serious or life threatening. The team wants to explore why the side effects are a marker for the success of the treatment.