A slightly updated version of a cellular immunotherapy that identifies mutations in a tumor of a patient has paved way to the full relapse of metastatic breast cancer that failed to react to various lines of chemotherapy.
This cellular immunotherapy is currently undergoing testing at the National Institutes of Health in Maryland in a Phase 2 clinical trial (NCT01174121). They’re also inviting patients with different kinds of advanced cancers.
The results of the study were published in the Nature Medicine journal entitled “Immune recognition of somatic mutations leading to complete durable regression in metastatic breast cancer.”
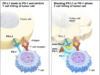
A personalized cancer therapy called autologous adoptive cell transfer utilizes the patient’s own immune cells, which are refined and expanded outside the body and is then introduced again to the patient.
Cellular immunotherapy has shown promising and positive results in high burden of mutations in cancers like melanoma. However, the therapy has lesser positive outcomes with lower mutation levels like epithelial cancers. Epithelial cancers includes ovarian, esophageal, stomach, and breast cancers.

Despite the several rounds of chemotherapy and hormonal therapy, the 49-year old patient with breast cancer failed to respond when she joined the clinical trial’s Phase 2, which happened at the National Health Institute. The process included DNA and RNA extraction from one tumor on the patient’s right breast and from the normal tissue. After this, the researchers then screened the genetic material for mutations that were distinct to her cancer. Researchers were able to extract immune cells from her tumor after detecting sixty-two (62) mutations in her tumor cells and determining the corresponding proteins. The extraction process is also known tumor infiltrating lymphocytes (TILs)m which enables researchers to see if the patient’s immune cells could distinguish any of the mutated proteins.
Subsets of her immune cells were found and were unique to four of these proteins. Researchers then expanded these subsets in the lab and administered them back into her bloodstream along with Keytruda (pembrolizumab, by Merck) to avoid immune cell inactivation.
The study reports that in total, she received 80,000 million cells that were given in 7 injections. In a span 6 weeks after her last infusion, it was discovered that the tumor size decreased by 51%, and that it continue to recede until no more remnants of her cancer were detected on imaging scans. This was comprised of her breast tumor and metastatic lesions in the liver and chest. For almost 2 years, the cancer regression is currently ongoing.
According to the National Cancer Institute (NCI)Center for Cancer Research (CCR)’s chief of the surgery branch, Steven A. Rosenberg, MD, PhD, they have developed a high-throughput method of determining the mutations found in a cancer that the immune system recognizes. Dr. Rosenberg also serves as the study’s lead author. He also added that despite the research being currently experimental, this new approach to immunotherapy is dependent on mutations and not on cancer type which can serve as a blueprint, in a way, and can be used for the treatment of various types of cancer.
It was also reported that patients with other epithelial cancers, including colorectal and liver cancer, had similar promising outcomes. Since all cancers have mutations, the goal of this immunotherapy is to attack these mutations, which ironically enough are the mutations that cause the cancer, but may prove to be the best bet to beating cancer.