Last May, President Trump passed a federal law that says terminally ill patients have the right to use experimental treatments that haven’t yet been fully approved by the Food and Drug Administration (FDA). This law, called Right to Try (RTT), will give patients with no other options the chance to extend their lives without having to wait for lengthy FDA approval processes.
Last May, President Trump passed a federal law that says terminally ill patients have the right to use experimental treatments that haven’t yet been fully approved by the Food and Drug Administration (FDA). This law, called Right to Try (RTT), will give patients with no other options the chance to extend their lives without having to wait for lengthy FDA approval processes.
“People who are terminally ill should not have to go from country to country to seek a cure,” said President Trump. “I want to give them a chance right here at home.”
So how do you know if you qualify for RTT treatments?
Well, first off, you need to have been diagnosed with a life-threatening disease or condition. According to Federal law, a life-threatening disease or condition is one “where the likelihood of death is high unless the course of disease is interrupted.” Diseases commonly treated by RTT investigational treatments include cancer and degenerative neuromuscular disease.
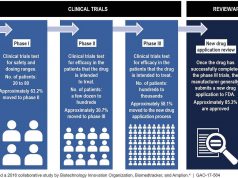
RTT treatments are intended to be last-line treatments that give some hope to patients who otherwise have no options. Thus, in order to qualify for RTT, you must have exhausted all of the treatments currently available to you. Many patients who turn to RTT treatments have not responded to prior treatments and are too sick to enroll in clinical trials. Participants of RTT cannot be eligible to take part in a clinical trial involving the drug they want to try. This information must be certified by a doctor who is in good standing with their licensing organization and will not be compensated by the drug manufacturer.
It’s important to note that the treatments available through RTT are investigational. While they have gone through a preliminary trial to determine initial safety concerns, the procedures are still associated with risks. You must give written informed consent that you know the risks associated with the treatment you want to try.