A clinical trial recently reported in the Journal of Clinical Oncology showed that several people with advanced Merkel cell carcinoma, a rare form of skin cancer, are responding remarkably well to immunotherapy.
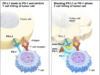
 The trial was conducted in 13 site around the country through the Cancer Immunotherapy Trials Network. The researchers used a checkpoint inhibitor drug called pembrolizumab, marketed as Keytruda. Checkpoint inhibitor drugs help the body’s immune system better target cancerous cells. Malignant tumors usually find ways to evade the immune system’s detection, but checkpoint inhibitor drugs “block” their ability to do that, which leaves the tumor wide open for attack. Keytruda is the treatment used by former president Jimmy Carter to treat his skin cancer, and the drug is now a standard treatment for many different cancers.
The trial was conducted in 13 site around the country through the Cancer Immunotherapy Trials Network. The researchers used a checkpoint inhibitor drug called pembrolizumab, marketed as Keytruda. Checkpoint inhibitor drugs help the body’s immune system better target cancerous cells. Malignant tumors usually find ways to evade the immune system’s detection, but checkpoint inhibitor drugs “block” their ability to do that, which leaves the tumor wide open for attack. Keytruda is the treatment used by former president Jimmy Carter to treat his skin cancer, and the drug is now a standard treatment for many different cancers.
In the past, the standard treatment for patients with late-stage Merkel cell carcinoma (MCC) was chemotherapy, but nearly 75% of those patients would die within two years.
 The clinical trial showed that after receiving pembrolizumab, 28 out of 50 patients saw their tumors either reduce in size or disappear completely. Nearly all of these patients have sustained these results for two years after their original treatment. The research team thus believes there is at least a 2-year survival rate of more than 68% for late-stage MCC patients treated with pembrolizumab.
The clinical trial showed that after receiving pembrolizumab, 28 out of 50 patients saw their tumors either reduce in size or disappear completely. Nearly all of these patients have sustained these results for two years after their original treatment. The research team thus believes there is at least a 2-year survival rate of more than 68% for late-stage MCC patients treated with pembrolizumab.
According to UW Medicine Newsroom, the leaders of this study have been very enthusiastic about its results. Martin “Mac” Cheever of Fred Hutchinson Research Center says that the high response rate to the drug is not very typical of solid-tumor cancers.
Both the trial and the drug have given many cancer patients hope where there previously was none. But there are still many studies to be done. The drug still does come with some risks, as a few patients experienced severe side effects, and one died. Cheever says the study is “the beginning, not the end.”
Researchers will continue to develop the treatment, and this study will help to guide future trials focused on treating MCC, which in the past, has been largely under-considered.
But for now, those with MCC can access pembrolizumab, as the U.S. Food and Drug Administration (FDA) approved it for the disease in 2018 as a result of this trial’s efficacy.